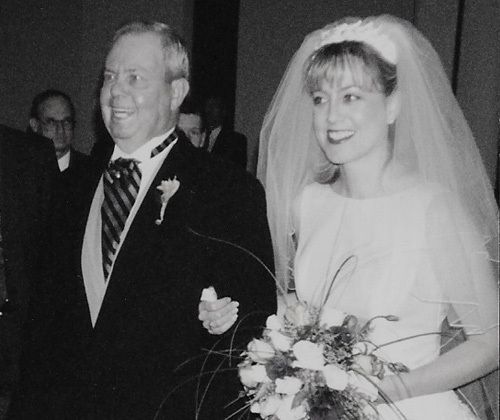Who We Are
The Kennedy’s Disease Association (KDA) is 501(c)(3) non-profit organization dedicated to improving the lives of those affected by Kennedy’s Disease (KD). For 25 years, the KDA has united patients, families, researchers, and advocates to share hope, accelerate research, and support one another on the journey toward a cure. Our mission has remained the same: to inform, support, educate, fund research, and ultimately find a cure for Kennedy’s Disease.
Featured Stories
The remarkable life and legacy of Dr. William Kennedy, the neurologist who first identified Kennedy’s Disease. Get to know the man behind the name and the lasting impact of his groundbreaking work.
For years, I believed my shaking hands were just an essential tremor – until I learned they were early signs of Kennedy’s Disease. In this deeply personal story, I share how KD has changed my life, and why I'm supporting the Kennedy’s Disease Association.
On my wedding day, my dad walked me down the aisle - a moment I wasn’t sure would be possible. He was struggling to stay on his feet due to what we didn’t know was Kennedy’s Disease.
News & Events

Join us for the upcoming KDA Fundraising & Volunteer Committee Meeting - it's an important monthly meeting to connect, collaborate, and contribute to our mission of finding a cure for Kennedy’s Disease. Our dedicated volunteers gather each month to plan fundraising initiatives, share updates, and explore new ways to support the KDA. Please contact us if you'd like to get involved. Together, we can make a meaningful difference.
Frequently Asked Questions
Have questions about Kennedy’s Disease? You’re in the right place. We’ve compiled answers to the most common questions here in our FAQ section.
-
What is Kennedy's Disease
Kennedy’s Disease (KD) - also known as Spinal and Bulbar Muscular Atrophy (SBMA) - is a rare, inherited neuromuscular disorder. It is also referred to as X-linked recessive bulbospinal neuropathy or X-linked spinal and bulbar atrophy.
KD is an adult onset, progressive disorder, characterized by the degeneration of lower motor neurons within the spinal cord and brainstem. This causes progressive weakening and wasting of the muscles particularly in the arms and legs. Please read What is Kennedy's Disease to learn more about the disease.
-
Is there a treatment or cure for Kennedy's Disease?
Unfortunately, there is no known treatment or cure, but researchers continue to make strides every year. Some individuals living with KD do take medications prescribed by their medical doctor to help alleviate various symptoms. Others have reported that a smart (light) exercise program coupled with stretching helps. If you have questions, visit the KDA Forum or contact the KDA.
-
What causes the symptoms that are associated with Kennedy's Disease?
Kennedy's Disease is a motor neuron disease, which involves the gradual degeneration of nerve cells responsible for controlling voluntary muscle movement.
Motor neurons are long nerve cells that extend from your spinal cord to your muscles. These nerve cells fire to make your muscles contract. In KD, the nerve cells become dysfunctional and eventually die, leaving the muscles unable to contract.
The androgen receptor is a protein that resides inside the nerve cell. Many cells have the androgen receptor protein, but motor neurons have more than most. The binding of testosterone to an androgen receptor somehow causes the onset of the disease. Men are generally affected by KD because they have much higher levels of testosterone than women.
-
 Len Janicki * NV, USA
Len Janicki * NV, USAIt was a relief to know that I was not alone. Now, we all have the KDA.
-
 Roger Stoufer * MN, USA
Roger Stoufer * MN, USAWe attended our first Kennedy Disease Conference in 2006 in Atlanta, Georgia. We have found hope, strength and encouragement through it. We are eternally thankful for everyone's support and participation.
-
 Ronni Bartlett * MN, USA
Ronni Bartlett * MN, USAThank you to the Kennedy’s Disease Association for being here to help support those living with Kennedy’s Disease.
-
 Mosie M. * AUSTRALIA
Mosie M. * AUSTRALIAThe KDA website was an absolute lifesaver for me, and discussions I had at various times have kept me going when I thought all was lost.
-
 Ted Abbott * CA, USA
Ted Abbott * CA, USAThanks for the Kennedy's Disease Association web site and the chance for those of us with Kennedy's Disease from around the world to share our experiences.
-
 Claus-Peter Czaya * GERMANY
Claus-Peter Czaya * GERMANYLife with SBMA remains worth living.
-
 Fernando Gines Ortega * SPAIN
Fernando Gines Ortega * SPAINThanks to the Kennedy's Disease Association, I have been able to meet some people affected by Kennedy's Disease.
Trusted Worldwide by the Kennedy's Disease Community
Proudly Recognized for Transparency, Accountability, and Earning Your Trust
-
Candid Platinum Transparency Seal2025 Candid Platinum Transparency Badge